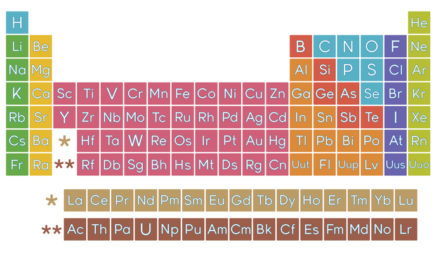
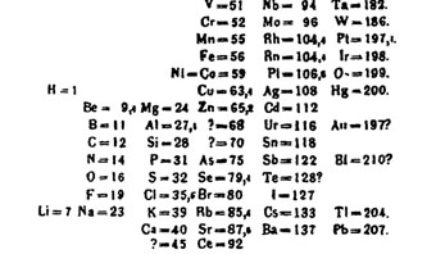Chemistry: Challenges and Solutions
The Reactivity of Alkali Metals Explained (animation)
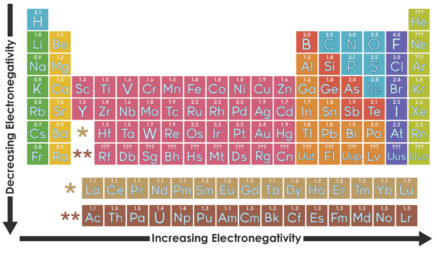
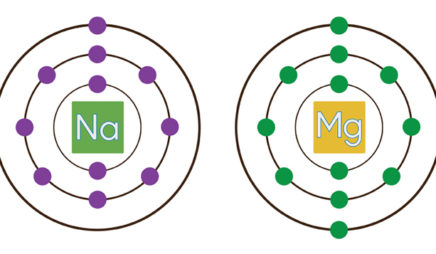
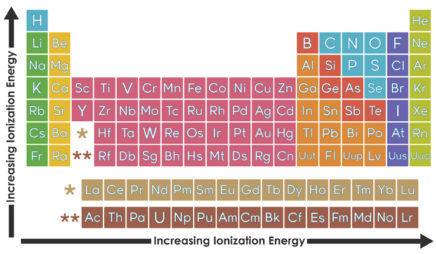
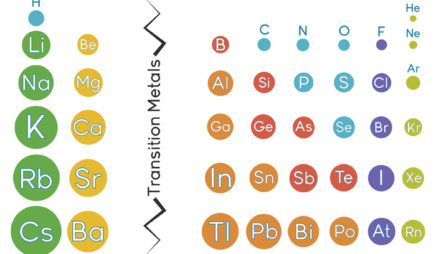
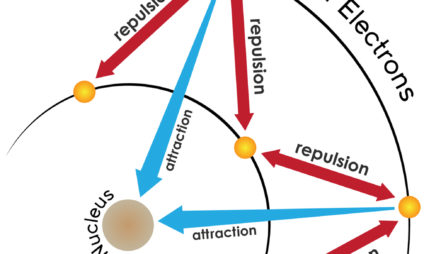
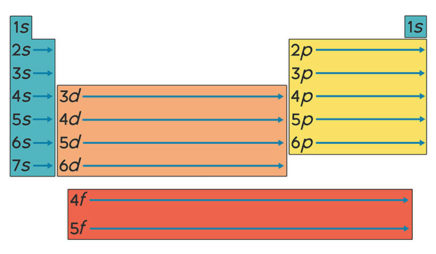
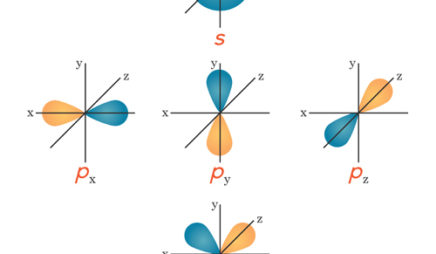
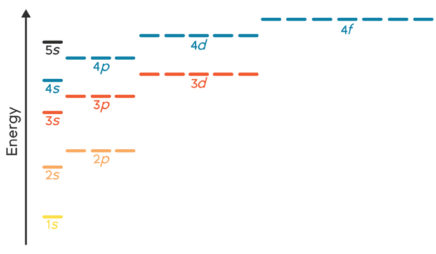
All the alkali metals—lithium, sodium, potassium, and so on—have only one electron in their valence shell. Because this one electron is likely to be far from the nucleus, it feels little attraction to the atom. The result: Alkali metals tend to lose this electron when they participate in reactions. These elements all “look” similar from the perspective of a reacting partner—the only difference is which shell is the valence shell.