Join us for conversations that inspire, recognize, and encourage innovation and best practices in the education profession.
Available on Apple Podcasts, Spotify, Google Podcasts, and more.
Look for the following topics in the video, indicated by the ![]() onscreen icon, and click below to learn more.
onscreen icon, and click below to learn more.
 Chemical Vs. Physical Change |
 Chemical Reactions in the Video |
 Are All Atoms the Same? |
 Where Does the “Weight” Go? |

Ice melting: an example of physical change.
Although some extensive properties (like shape, phase, etc.) of the material change, the material itself is the same before and after the change. The change can be “undone.”

A nail rusting: an example of chemical change.
The substances present at the beginning of the change are not present at the end; new substances are formed. The change cannot be “undone.”
Mass is neither created nor destroyed.
What are the macroscopic and microscopic differences between physical and chemical changes?

Many changes of state are easy to characterize based on the definitions above, including the following:
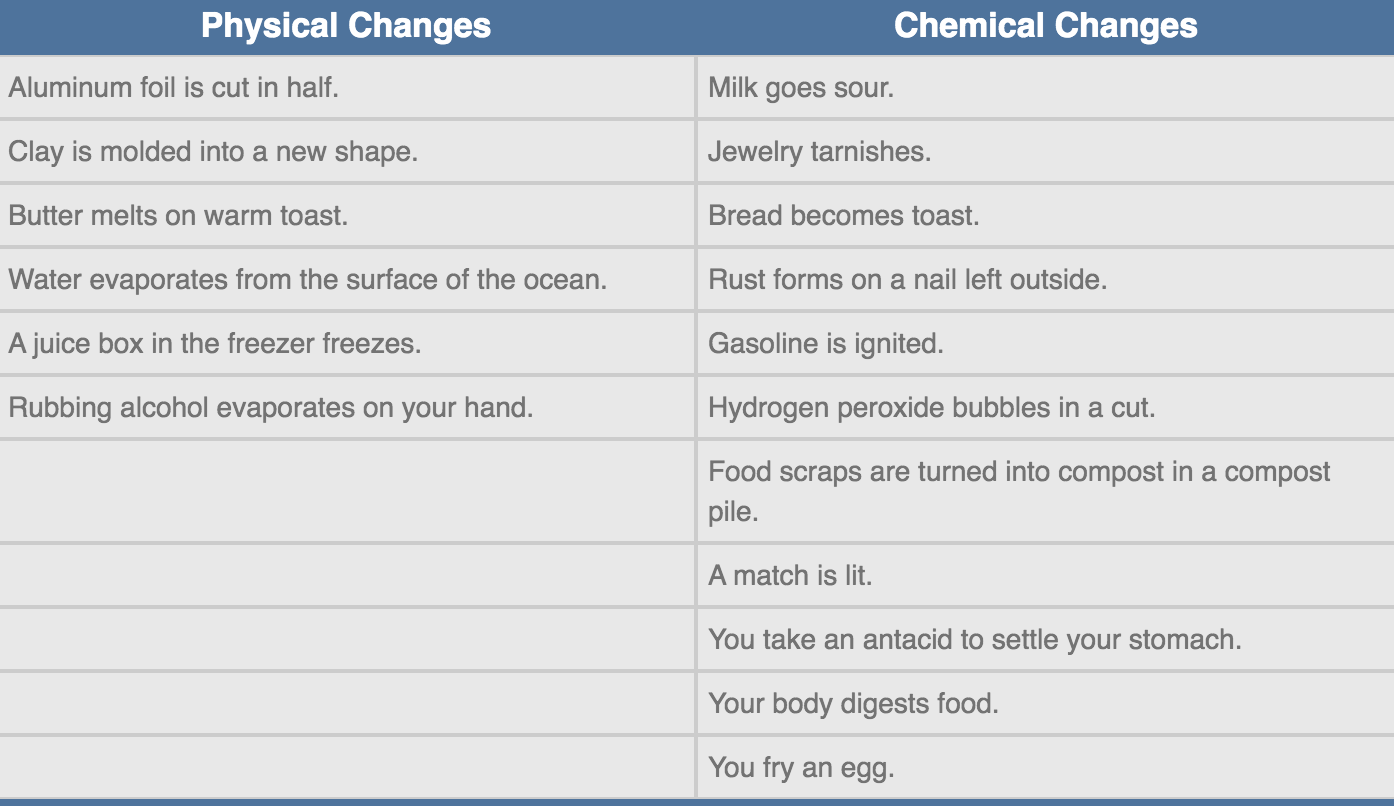
There are many cases where the distinctions between physical changes and chemical changes are unclear. For example:
The dissolution of salt in water: This seems like a physical change because we know we can recover the salt from the water. However, if we look at the microscopic level, we see that the two types of atoms in salt, sodium and chlorine, separate from one another. In this example, we don’t have a new substance, therefore this salt in solution doesn’t fit the microscopic definition of a chemical change; but we also don’t have the substance in its original form — a stack of alternating sodium and chlorine atoms. Does this mean the change is half chemical and half physical? Though it has aspects of a chemical change, scientists would still classify the dissolution of salt as a physical change.
The creation of a metal alloy: If we melt two types of metal together, we create an alloy metal that has different properties than either of its components (e.g., heat conductivity, electrical conductivity, density, etc.). This might lead us to think that we’ve witnessed a chemical change. In fact, a new particle is not created by melting two metals together. This indicates they did not undergo a chemical reaction. Brass, for example, is about 60% copper and 40% zinc, and is composed of individual copper and zinc atoms (i.e., there is no “smallest unit” that is still brass). There is no such thing as a brass molecule.
The heating and cooling of certain rubbers and plastics: One might think that exposing certain rubbers and plastics to heat or cold would cause a chemical reaction because the properties change (e.g., the materials become more rigid and brittle). While chemical reactions do take place, they simply bind together different parts of the large molecules that compose rubber and plastic. These new bonds add to the rigidity of the material, but the particles of the substances remain the same.
Chemical reactions can be very different from one another, giving off different gases, different amounts of heat, and even different smells. At a microscopic scale, hundreds of thousands or even millions of encounters between pairs of particles are responsible for creating the macroscopic changes that we can observe. (Not surprisingly, Dalton, the 18th century English scientist, often called “the father of modern chemistry,” referred to this process as the “dance of the atoms.”) The key to a chemical reaction is that the attractive forces between different particles are strong enough to tear the particles apart from each other.
Molecules that have two or more atoms are called compounds. These compounds are held together by the forces between their component atoms and/or ions, which are atoms with either an excess or deficit of electrons. When we mix two different compounds together (the “reactants”), the molecules of each will be composed of two or more different atoms or ions.
If when the molecules of the two compounds collide, they are in contact for only a split second, they simply bounce off each other, no reaction takes place, and the original materials remain. However, if during a collision, an atom or ion of one molecule feels a force from one of the atoms/ions in a second molecule that is stronger than the force that held it to its original molecule, it will combine with the more attractive atom/ion, forming a new molecule (the “product”). At this point, a chemical reaction has occurred.
This process is also consistent with the principle of conservation of matter presented in Session 3: atoms and ions can rearrange into different molecules, but they never just disappear. As a result, in a chemical change the total number of particles is not changed. Our microscopic definition of chemical change is now, as our host Sallie Baliunas states in the video: “a dissociation, recombination, or rearrangement of [particles].”
1. Lye (drain cleaner) and Epsom salt
Sodium hydroxide + magnesium sulfate —-> magnesium hydroxide and sodium sulfate
2(NaOH) + MgSO_4 —–> Mg(OH)_2 + Na_2SO_4)

We begin by dissolving the compound Epsom salts in water, thereby splitting the molecule into its component atoms and ions.
The second compound, the lye (or liquid drain cleaner) is already dissolved in water.
When these two reactants are mixed together, the ions that make up the sodium hydroxide and magnesium sulfate collide. Because the forces attracting these particles to each other are greater than the forces between them and the water molecules, new molecules of magnesium hydroxide and sodium sulfate are formed. We see white products “precipitate” out of the clear mixture and the solution that remains is different from either of the original reactants:

2. Baking soda and Vinegar
Sodium bicarbonate + acetic acid gives sodium acetate, water, and carbon dioxide.
NaHCO3 + HOOCCH3 ——> NaOOCCH3 + H2O + CO2 )
In this reaction, we mix the baking soda (sodium bicarbonate), a solid powder, with vinegar.


When we bring these compounds together, two baking soda particles and one vinegar particle collide. In this case, the attractive forces between two reactants come together and three products are created, a molecule of sodium acetate that remains in solution, a water molecule, and a molecule of carbon dioxide. Carbon dioxide is a gas at room temperature, so it bubbles up out of the mixture as the reaction takes place.


John Dalton.
Although Dalton was able to make correct predictions about the phenomena he observed in the 1800s, one of his main assumptions — that all atoms of a given substance are identical — was not correct, based on what we now know about the subatomic structure of the atom.
While an atom is the smallest particle of an element that retains the intensive properties of that element, all 92 stable atoms are made of only three smaller, more fundamental particles: the proton, the electron, and the neutron. A simple model of the atom (refined by physicist Neils Bohr in 1922) shows the protons and neutrons packed together in the core of the atom, called the nucleus, and the electrons orbiting in specific paths around the nucleus. Although the development of quantum mechanics in the early 20th century led to our modern understanding of the atom as having a more indefinite structure than Bohr described, most chemical interactions among atoms are governed by the electrical charges of their electrons and protons.
Since Dalton was only able to probe the chemical behavior of materials, he assumed all atoms of the same element were identical. Recall that neutrons, along with protons, make up the nucleus. Since neutrons have no electrical charge, they do not affect the chemical behavior. What Dalton didn’t know is that there can be different numbers of neutrons in atoms of the same element — that is, the atoms are not all identical. Atoms of the same element with different numbers of neutrons are called isotopes.
Although chemical behavior is not different for most isotopes, the behavior of different isotopes of the same element at very high temperatures and pressures can vary. Nuclear fission reactions, which occur in nuclear power plants and stars, offer a good example of ways the isotopes can affect an atom. For example, when a uranium atom with 92 protons and 143 neutrons (U^235) is bombarded with neutrons, it releases large amounts of energy in a fission reaction. However, a uranium atom with 92 protons and 146 neutrons (U^238) will not produce a fission reaction when bombarded with neutrons.

“Cheese” candle.
Is burning a candle a good analogy for what happens to food inside the body? During the video, Robin and Sallie, our hosts, burned a “cheese” candle as a possible model for what happens to food inside our bodies. Although it is true that food is essentially the fuel for our bodies, the process by which we extract energy from it is different than the process by which a candle burns.
When a wax candle is burning, its particles, made mostly of carbon and hydrogen atoms, combine with oxygen in the air to produce carbon dioxide and water vapor. In addition, some energy is released in the form of heat. (We’ll learn more about heat in Session 7.)
However, our bodies do not “burn” particles of food in a single reaction like the burning candle). Instead, a series of reactions takes place during which the result of one reaction becomes the input to the next. Food is broken down into simple sugars made mostly of carbon, nitrogen, oxygen, and hydrogen molecules that are then combined with the oxygen we breathe. Along the way, other molecules (called enzymes) help the reactions occur.