Join us for conversations that inspire, recognize, and encourage innovation and best practices in the education profession.
Available on Apple Podcasts, Spotify, Google Podcasts, and more.

Cells

Organic Molecules

Extremophiles

Teaching Tips

Scientist at Work
Curriculum Resources
In 1665, a physicist named Robert Hooke used one of the first microscopes to look more closely at the living world. A slice of cork caught his eye. Looking at thousands of tiny chambers, Hooke termed these structures cells because they reminded him of the rooms in a monastery.
Subsequent observations built upon Hooke’s work and led to what is known as the cell theory. The cell theory states the following:

Hooke’s microscope.

Cork cells as seen in an early microscope.
An organism may be characterized as having different levels of organization built around the cell. Unicellular organisms are made up of only one cell, while some multicellular organisms are comprised of trillions of cells. In multicellular organisms, groups of cells sharing similar structure form tissues that carry out a specialized function. Different tissue layers form organs. And separate organs often work together to carry out major bodily functions, such as digestion or circulation. These “teams” of organs are referred to as organ systems. Growth of an organism is the result of an increase in cell size and, in multicellular organisms, an increase in the number of cells.
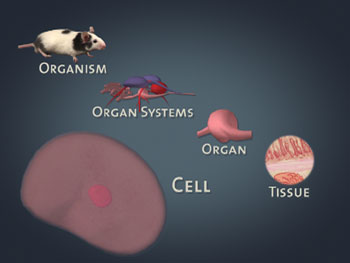
Levels of organization.
At first it might seem that the cells of such dissimilar creatures as the tiniest bacterium and a great blue whale would be very different. The cells of any organism, however, demonstrate all the characteristics of life: a life span that includes a living beginning, growth, development, reproduction, and death; the constant use of matter and energy; response to changing internal and external conditions; and the presence of DNA.
Although multicellular organisms can have diverse cell types, all cells have certain parts in common (see the interactive cell picture below). All cells are bound by a cell membrane and contain cytoplasm, which is where the cell parts lie. The nucleus of this cell contains the hereditary molecule DNA, which is also found in all cells. And, all cells contain ribosomes, which are so tiny that they can’t be seen in this cell. Ribosomes are structures that are responsible for constructing proteins—the molecules that carry out the instructions contained in DNA.
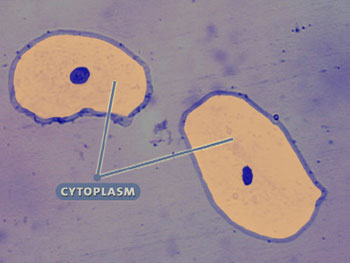
Cytoplasm
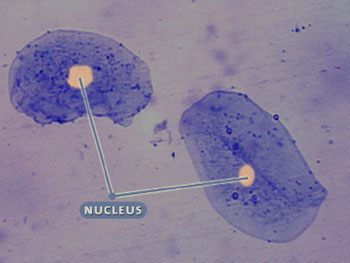
Nucleus
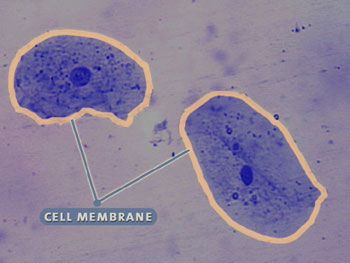
Cell Membrane
One of the most basic distinctions among life forms involves the cell. In some cells, including those of plants, animals, fungi, and protists, the DNA is contained in a nucleus, which is itself a membrane-enclosed body. Cells that contain a nucleus are called eukaryotic (“true nucleus”) cells, and the organisms with cells of this type are called eukaryotes. Within the cytoplasm of a eukaryotic cell, there are numerous compartmentalized bodies called organelles. Organelles serve specific purposes within the cell, ranging from building proteins to making energy. Different types of organisms may have different sets of organelles. Organisms that photosynthesize, like plants, contain chloroplasts, for example.
There are cells that aren’t as structurally complex as those with a nucleus. Prokaryotic (“before the nucleus”) cells lack a nucleus and organelles. Most single-celled organisms are prokaryotes that are known as bacteria and archaea. Prokaryotes are still capable of performing many of the same types of functions as eukaryotes. These functions occur in the cytoplasm instead of in organelles. In fact, many bacterial and archaeal cells are capable of carrying out very complicated biochemical reactions that allow them to harvest food and/or energy from very unlikely sources, such as pollutants and poisons.

Organic Molecule
Like all matter, life is built from atoms — the basic units of matter that link together to form molecules. Organic molecules are the molecules of life and are built around chains of carbon atoms that are often quite long. There are four main groups of organic molecules that combine to build cells and their parts: carbohydrates, proteins, lipids, and nucleic acids.
Each carbon atom can form strong, stable bonds with four other atoms at a time – these are usually oxygen, hydrogen, nitrogen, sulfur, and phosphorus atoms. Carbon can also bond to other carbon atoms to form chains that are almost unlimited in length. This creates a huge number and variety of molecules that can be built from carbon atoms. No other element even comes close. Organic chemistry is the study of these carbon-based molecules.
In addition to composing living matter, organic molecules are found in the nonliving world. Fossil fuels, which are derived from the buried remains of once-living organisms, are made of organic molecules. So are many man-made compounds, such as plastics, pesticides, and many medicines. The clothes we wear are all made of organic molecules: wool, silk, cotton, polyester, nylon, acrylic, and rayon. Many household and personal goods are organic as well, including detergent, cleaners, soap, shampoo, and perfume.
Although there are some exceptions, most naturally occurring nonliving things are made of inorganic molecules. Inorganic molecules are not composed of chains of carbon atoms and are generally much less complex. Living things do use inorganic molecules—like water, oxygen, and carbon dioxide—and their atoms may even be assimilated into organic molecules. Plants, for example, use carbon dioxide with the carbon being assimilated into a carbohydrate: sugar.

Thermal vent
One of the great thrills of microbiology is studying organisms that thrive in some of the world’s most inhospitable environments. These organisms are known as extremophiles (“phile” means “loving”). They live in extreme environments and are able to exploit resources that are unusable, and many times toxic, to other organisms.
The vocabulary of extremophiles describes the extremophiles themselves. It’s actually very straightforward and based on interchangeable prefixes and suffixes:
| Acido- | Acidic, low pH conditions |
| Alkali- | Alkaline, high pH conditions |
| Halo- | High salt conditions |
| Psychro- | Cold temperature |
| Thermo- | Hot temperature |
| -phile | “loving” |
There is even a distinction made for microbes found in the most extreme environments. For example, hyperthermophiles live in environments hotter than 80ºC (“hyper” means “above”). There are even organisms that are able to exist in acidic hot springs, making them acidothermophilic.
While extremophiles are intrinsically interesting because they can inhabit otherwise barren niches, there are also benefits to humans. For instance, the key enzymes used by molecular biologists to examine DNA sequences were isolated from thermophilic bacteria. These enzymes are naturally able to withstand the high temperatures necessary for scientists to manipulate DNA. There are also extremophile enzymes that are well suited for industrial applications that are carried out at elevated temperatures or in acidic or alkaline conditions. Extremophiles are constantly under examination by researchers for additional agents that could facilitate academic, industrial, and even medical projects.
 What’s on the menu at the SPONCH CaFe? The elements of life! Our host Dr. Douglas Zook provides us with a clever way of remembering some of the most common types of elements found as atoms in the molecules of living things. This mnemonic device is based on the chemical symbols for elements in the periodic table.
What’s on the menu at the SPONCH CaFe? The elements of life! Our host Dr. Douglas Zook provides us with a clever way of remembering some of the most common types of elements found as atoms in the molecules of living things. This mnemonic device is based on the chemical symbols for elements in the periodic table.
The elements sulfur (S), phosphorus (P), oxygen (O), nitrogen (N), carbon (C), and hydrogen (H) are the most abundant in organic molecules, but calcium (Ca) and iron (Fe) are also critical to life functions. Dr. Zook even goes further with his students by adding “Maggie’s SPONCH CaFe”, where Maggie stands for magnesium (Mg). Of the 107 elements that occur naturally on Earth, only about a dozen or so compose most of the matter of an organism. There are many other elements, however, that are very important and occur in trace amounts. Furthermore, different types of organisms often have different proportions of these elements.
 Dr. Gary Ruvkun is a professor of genetics at Harvard University. He is the head of a large research lab in the department of molecular biology at Massachusetts General Hospital. His research group focuses on a diverse set of questions pertaining to the molecules of life.
Dr. Gary Ruvkun is a professor of genetics at Harvard University. He is the head of a large research lab in the department of molecular biology at Massachusetts General Hospital. His research group focuses on a diverse set of questions pertaining to the molecules of life.
One prominent project involves the search for life on Mars. Mars has an inhospitable environment that nonetheless shares certain features with the Earth — most markedly water and certain types of landforms. Dr. Ruvkun proposes to look for life on Mars by taking sediment samples and testing them for the presence of DNA — the hereditary material of life on Earth. Finding DNA on Mars would provide evidence that there are — or were — life forms on Mars related to those on Earth. Which planet harbored these creatures first would remain a question.
SCIS 3+ consists of 13 units, and spans from kindergarten through sixth grade. The concepts developed in each unit of SCIS 3+ are elaborated upon throughout the program. Students acquire a strong background for building scientific literacy, which enables them to make intelligent decisions about their environment and the world in which they live.
The original program, SCIS, integrated the learning theories of Jean Piaget. Using the Learning Cycle strategy, students are led to discover concepts through their investigations with a wide variety of manipulative materials and live organisms. Completely activity-based, the SCIS 3+ approach is based upon current research on how children learn. The SCIS approach to teaching science concepts and process skills has been proven through years of classroom use.
SCIS 3+ is a revision of the work of the Science Curriculum Improvement Study, which was developed by the Physics Department and the Lawrence Hall of Science of the University of California, Berkeley, with the support of the National Science Foundation. Development of SCIS 3+ was supported and copyrighted by Delta Education. For more information, contact:
Delta Education, Inc.
phone: 1-800-258-1302
https://www.deltaeducation.com/