|
|
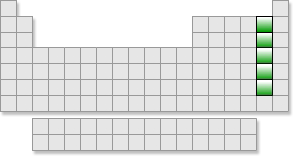
Groups :Halogens |
About the Group Group 17 is a family of elements known as the halogens. The word "halogen" means "salt-former." The halogens form salts when they react with a metal. Halogens exist in a variety of states at room temperature. Fluorine and chlorine are gases, bromine is a liquid, and iodine and astatine are solids. All of these elements exist as diatomic molecules in their gaseous state. This means that two atoms bond together to form a molecule of the gas. Chemical Properties The elements in this group are all one electron short of having a complete s-p octet in their outer energy level. Any element that accepts electrons to form a complete octet is called a nonmetal. The halogens will typically take an electron from a metal or even a weaker nonmetal to complete the desired octet. The ions formed by this transfer of electrons to the nonmetal are negatively charged because they have more electrons than protons. They are called anions. Fluorine is the element with the most electronegativity on the entire periodic table. Electronegativity is the measure of how strongly an element will attract electrons away from other atoms. Its very high electronegativity makes fluorine the most reactive element. In the "Real" World All of the elements of the halogen family are found in common use in everyday life. Fluorine is used in compounds to strengthen the enamel of your teeth against decay. It is also used in acid form to etch glass. Chlorine is used in our drinking water and in swimming pools to inhibit bacterial growth. It is also used in the form of chlorine dioxide to bleach wood pulp in the manufacture of very white paper. We also use chlorine in everyday laundry bleach. Chlorine compounds are used in insecticides, fireworks, and matches. |
 |
|||
| Next: Oxygen Family |