Rediscovering Biology: Molecular to Global Perspectives
Cell Biology and Cancer Expert Interview Transcript: Mary-Claire King, Ph.D.
 Professor of Medicine and Genetics
Professor of Medicine and Genetics
King is an American Cancer Society professor of medicine and genetics at the University of Washington in Seattle. She was the first to prove that breast cancer is inherited in some families. She is now investigating the genes that predispose some women to breast cancer to learn what these genes may reveal about breast cancer generally.
Interview Transcript
Interview with Mary-Claire King, Ph.D. King is an American Cancer Society Professor of Medicine and Genetics at the University of Washington in Seattle. She was the first to prove that breast cancer is inherited in some families. She is now investigating the genes that predispose some women to breast cancer to learn what these genes may reveal about breast cancer generally.
Since 1970 to present-day what have we learned about cancer?
When those of us who are now middle-aged went to high school and to college, what we learned about cancer was completely descriptive. We learned how cancer cells look compared to the way normal cells look and it was beautiful, it was elegant. We learned how cancerous organs look compared to the way normal organs look. We learned about how patients decline with cancer. But it was very frustrating at least for me, because we didn’t have any understanding or sense of why these processes were occurring. Exactly what was happening, why it was it happening, when was it happening, how was it happening, all the questions you ask of mystery. We now don’t have them all answered — if it were an easy problem it would have long since been solved. But we do have a very good sense of the kinds of changes that a cell undergoes between the time it is a normal cell and the time that it is growing completely out of control, causes a tumor that can invade, metastasize and kill its host.
If I had to say one thing about cancer is that there is an extraordinarily wide range of alterations that can occur, of pathways that can be abrogated. We’ve learned that cancer is a very large number of different phenotypes of different traits. The single most positive thing that we’ve learned about cancer is that we now know that it is genetic. Cancer is always genetic. It’s always caused by changes in the DNA, always caused by mutation. The number of genes that can be mutated between the time that a cell is dividing normally and the time its cell division is out of control are extremely numerous.
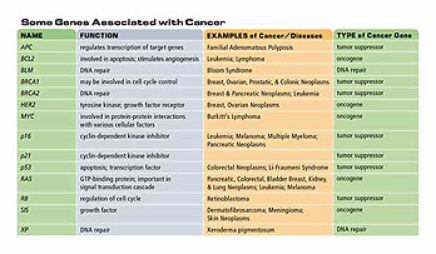
There are some major [gene] players — p53, RB — there are some players that are major in one organ system and not another. But these players, these genes that undergo mutation in cancer cells, tend to be in pathways that are critical to the normal, healthy division of cells. So they are mutations in genes and pathways that repair DNA; they are in pathways that control the rate of cell division; they’re in pathways that control the normal process of cell death; and they’re in pathways that control the environment of the cell. When you think about it, it is extraordinary that cancer is kept under control by homeostasis in the body to the degree that it is, because there are a lot of things that can go wrong.
So one of the very basic things that we are increasingly aware of now is how early mutations in cancer are very often in those genes that preclude other mutations from happening. Mutations in genes that are critical to DNA repair are often the first step in what then becomes a series of mutations, almost a cascade of mutations that leads to cancer.
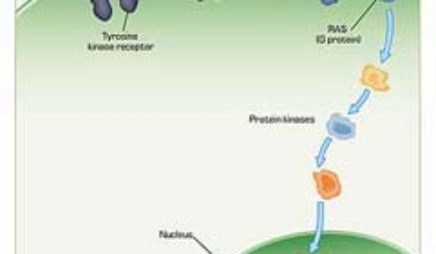
But let me back up a bit. I asserted that cancer is always genetic, and back in 1970 that would have been heretical to say. The role that the early, brilliant workers thought that viruses played in cancer turns out to be the role for human oncogenes; that is the human homologues of viral genes in other organisms indeed have a major role in cancer. Oncogenes are a major class of cancer genes. The nature of the aberrations in oncogenes that lead to cancer is [when] a gene — which in its normal manifestation permits a cell to grow at a nice, progressive pace-undergoes a mutation, which enables the cell to grow way too fast. The analogy has been drawn between having an oncogene being an accelerator on a car, which normally [is] regulated by pressing it just as much as you need it. A mutation in an oncogene that leads to cancer, [is] an aberration that keeps the accelerator permanently on. So the cell now grows out of control. Mike Bishop was the first to come up with [this analogy].
Besides oncogenes, what other mutations can lead to a cancerous cell?
A tumor suppressor gene is a gene that in its normal manifestation keeps cell division under control and it puts a break on the aberrant division of a cell. If it goes awry, that break is no longer present. Here the analogy is to the brakes on the car; so under normal circumstances the brake is present when you need it, you press on it gently and you slow the car.
Tumor suppressor genes are genes with a wide variety of normal functions in the cell. I’ve always felt a little queasy about the idea of calling these tumor suppressor genes because it suggests that the gene evolved in order to suppress tumors. But these genes didn’t evolve in order to suppress tumors. We and our neighbors the yeasts were around for a long time before there were tumors. These genes evolved to take care of the normal healthy life of cells. But they have the consequence that when their functions are completely abrogated, either as the result of mutation or a result of being down-regulated by some other process, the cell is no longer able to function in a normal controlled way and now grows out of control. The mechanisms by which various tumor suppressor genes fail to carry out that function, of course, are heterogeneous and it depends on the nature of the tumor suppressor gene.
My favorite tumor suppressor genes are BRCA1 and BRCA2, which are the genes which when inactivated lead to breast and ovarian cancer. These genes functionally have many different roles in the cell. BRCA1 acts in the maintenance of what we call genomic integrity. It acts in concert with many other gene products to preclude mutations that inevitably occur to all cells all the time. In other words, it acts with its colleagues in the cell to repair damaged DNA. It also regulates transcription of other genes which ought to be expressed at just the right levels.
So it works in a whole series of pathways. It has multiple functions and multiple protein partners. When [there is] either a mutation of BRCA1 in the breast duct, in the epithelial wall of the ovary, or as the consequence of a combination of inherited mutations, and there are no normal copies of the BRCA1 gene left, then the functions in which it participates fall apart for that cell. An individual, mouse or person, who has both copies of BRCA1 lost at conception dies very early in embryonic development. If there are no cells in the body that can repair DNA, that can get the right genes transcribed, and that can maintain genomic integrity, obviously, that is not compatible with life.
However, since each of us [has] two copies of every gene, one from our mother, one from our father, we have two copies of BRCA1 and two copies of BRCA2. To have one of those copies abrogated by mutation and therefore non-functional, in and of itself has no impact at all, all by itself. No problem. Indeed you will never meet a healthier group of people than the people with the inherited mutations in BRCA1 and BRCA2-unless somewhere in the breast ductile epithelium the second copy of BRCA1 goes bad. These mutations can be inherited either maternally or paternally, chromosome 17 for BRCA1, chromosome 13 for BRCA2. If I were a woman who inherited such a mutation I would be just fine. But all of my cells would have only one functional copy of BRCA1 left.
The danger, therefore, is that anytime a cell loses the one remaining normal copy of BRCA1, which is the consequence of a chance mutation, that cell no longer maintains it’s genomic integrity. This second loss is called somatic — a body-based hit. And that cell is now predisposed to go on and develop into a tumor. These tumors develop very fast. They quickly pick up other mutations. Generally in critical genes like p53 which has been called the guardian of the genome-a gene which when it goes awry really sets off a permissive cascade for other mutations to accumulate.
Can you explain the role of p53-why is it so important?
P53 is critical because it protects the cell from a variety of other kinds of mutations that can occur. Consequently, p53 mutations are shared by a wide variety of cancer types. Many of the tumor suppressor genes [are] altered only in colon cancer or only in breast cancer or only in endometrial cancer. P53 is altered in a wide variety of tumor types. We don’t yet know why p53 has such ubiquitous critical importance and why other tumor suppressors are more organ-specific, but it is certainly the case.
Mutations in p53 are almost all cell-specific — occasionally they are inherited. But mostly they are mutations that either abrogate the function of p53 entirely or alter its function. One of the first sorts of mutations that pathologists frequently find when they evaluate tumor tissue is a mutation in p53. Consequently, efforts to restore normal p53 function in tumors are an enormously important area of cancer research now.
The convergence of the availability of serious amounts of money in the ’70s, with the development of the new field of molecular biology and subsequently molecular genetics and subsequently genomics has really permitted the breakthroughs that we now savor in terms of trying to sort out what cancer is and what can be done about it. The nature of the experiments that we can undertake has changed radically just in my lifetime in science.
We can now expect to manipulate cells individually, obviously out of the body. We can expect to be able to evaluate protein by protein, gene by gene, and now base pair by base pair, all the genes that we are interested in. Name a gene, we can evaluate it. So the development in molecular biology and molecular genetics has enabled our study of cancer. It’s been a completely symbiotic relationship.
When Nixon first declared war on cancer, I was a young assistant professor at the time, and many of us were very cynical about it. We thought, “Oh he is just trying to throw money at a problem for a political reason.” Regardless of his motives, it worked. We know much more about cancer than we did before. There are now some sorts of cancer for which we have very good treatments that we didn’t have before.
Some forms of leukemia and lymphoma, Hodgkin’s disease, [colon cancer], and breast cancer have a much better survival than they used to have. There are cancers for which we have early detection schemes that we didn’t have before. So we have made progress in the practical areas related to cancer.
We’ve made an enormous amount of progress in the development of this field of molecular biology and molecular genetics that permits us to move forward quickly.
What happened in the understanding of cancer that allowed researchers to come develop a drug like Gleevec?
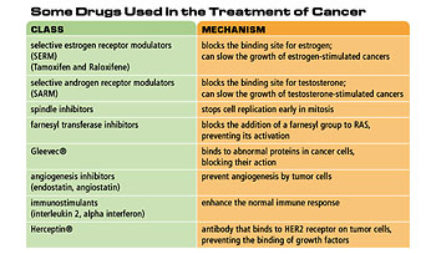
The development of drugs that are specific to certain sorts of cancer-indeed to certain genetic alterations in cancer-is specifically the result of being able to identify what those alterations are, and then to identify target compounds that can be used to preclude the consequences of those alterations. Finding the original genetic alteration is absolutely critical, but it is only the first step. Those of us who do genetics think of it as the Holy Grail. But the Holy Grail is only the Holy Grail if you figure out what to drink out of it and how to move forward with it. Once you know what the genetic alteration is, you have to then [ask] what is the consequence of this at the level of protein, what is the consequence of that at the level of the cell? And how can I develop a molecule that will preclude those consequences from permitting invasion of a cancer?
So that paradigm played out beautifully in the development of Gleevec; that is a model for the sort of a way it will play out for many drugs. Exactly how that works is a case-by-case story.
Can you talk about the multi-hit nature of cancer?
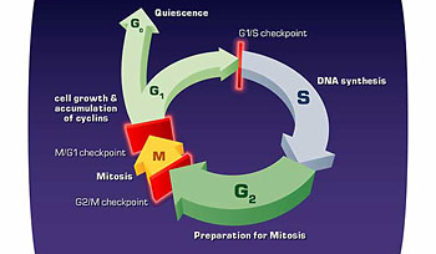
It would have been easy to solve cancer if cancer always developed in the same way. But of course, it doesn’t. If one evaluates a series of tumors, even of the same sort, [such as] a series of breast tumors from 20 different women, one will find mutations in different genes. There is an enormous variety of genes that can go wrong. We would like to have a complete catalog of what all those genes are, and we are moving right along in developing it. What we don’t yet know is what are all of the critical steps that can be rate-limiting in the development of a cancer. There have been some very good mathematical modeling done that predicts that the number of mutations that have to occur for a cell to go from being normal to being a cancer cell must be as many as 4, 5, or 6. But you say to yourself “that takes way too long, that takes hundreds of years.” How could that possibly happen?
We think now that the reason that a cell can accumulate so many mutations is that the first mutations that occur are frequently mutations that allow the cell to ratchet up the frequency of mutations that occur. So the first mutations that occur in carcinogenesis are very frequently mutations that kill off the process of normal repair of mutations. Because a lot of the cell’s effort is devoted to repairing errors, if that effort is no longer successful then every little thing that can go wrong does-you begin a downward spiral toward the development of cancer. Now from the point of view from the cell, that is a success because the cell has now made itself immortal. But from the point of view of the person, it’s a disaster.
Cancer seems to be a disease of mostly older people. Can you talk about that?
The reason that the major risk factor for most cancers is age is that it simply takes a while for mutations to accrue. If the probability that the mutation will occur is the same in every time interval, then the cumulative probability that a mutation will occur is obviously longer-the longer period of time and the older the individual. Why some cancers occur in young children is still partially a mystery. Although it is now clear that some cancers occur in young children because of inherited predisposition, for example, retinal blastoma (RB). [It was] discovered back in the late ’60s and early ’70s that children who had inherited one mutation in the RB tumor suppressor gene had both much earlier onset of retinal blastoma-typically it was congenital, they were born with it. Also, they were much more likely to have retinal blastoma in both eyes — that is the retinas of both eyes already [have] cancers in them.
Any cancer that is the consequence of both a cell-specific mutation and inherited mutation will occur earlier because one is born with one mutation, it doesn’t take any time at all to happen it’s already happened. And furthermore it’s already present in all the cells of the body. So that any cell in the target tissue is now vulnerable.
Is it worse to get a mutation or a tumor suppressor than an oncogene?
Mutations in either tumor suppressors or proto-oncogenes can be devastating. I don’t think there is a better or worse about it. Very frequently, a cancer upon evaluation with have mutations in both. Typically for mutation in tumor suppressor genes to cause cancer both copies have to be knocked out. Whereas mutations in oncogenes are primarily activating mutations, so there are mutations in the one copy of the oncogene that make it more active. Whereas mutations in tumor suppressor genes are typically mutations that wipe out the function of the tumor suppressor genes so it becomes inactive.
Can you expound on the idea that cancers are genetic but they are not all inherited?
We correctly think that all cancer is genetic, but only a very small fraction of cancer is inherited from parents to children, under 10% typically for most types of cancer.
That brings up the question, what is the difference between what’s inherited and what’s genetic. Indeed what is the difference between inherited, genetic and familial? So cancer is always genetic but it’s rarely inherited. Can you think of something that is inherited but not genetic? Money. So there is no earthly reason that a trait has to be both inherited and genetic, cancer sometimes is. But it’s always genetic.
So how does all the knowledge about cancer that we’ve gained in the past few decades help us with diagnosis and treatment?
What we’re trying to do now with the information we’re accumulating about how cells go wrong is to try to develop tests that pick up lesions extremely early. Imagine that an ovarian epithelial cell had gone bad and there were consequently alterations in its DNA, and the lesion was far too small to be detected yet, even by ultrasound. But [let’s say that] some cells of this very tiny proto-tumor had broken off and released their DNA into the peritoneal cavity. The DNA in and of itself wouldn’t have any consequence at all, it’s just DNA floating about. But if we had the capacity to detect [this DNA] from a peritoneal sample from a young woman, 35, 40 years old, we might be able to say “there is a p53 mutation somewhere in your ovary we need to have a look.” We could then get a very early sense that there was something wrong. If we could have a very high likelihood of this test being both sensitive and specific, it would be fabulous because we’d be able to pick up these tumors much earlier. In breast cancer, we could identify what the lesions were in breast epithelial cells that had consequences on the surface of the cells and then develop antibodies to whole repertoire of those potential lesions. [We would then] inject those antibodies into the blood stream-anything that goes into the bloodstream goes into the breast ducts. Those antibody labels [would] flow through the breast ducts and [we would] take a picture, the same way we do a mammogram now, but it’s a picture that will pick up an antibody hypothetically on the surface only of the breast cell that has gone awry.
[It would be] fabulous [to] have an early detection scheme that is cell-specific. We don’t have to wait for a lesion to be half a centimeter in size. By the time a tumor is that size, it has lived more than 80% of its life as a tumor. All of us in cancer biology, would like to push back early detection. So that invasive cancer is a thing of the past. I don’t think we will ever be able to completely prevent mutations from happening. But we should be able to prevent them from becoming tumors that kill their hosts.
At the level of treatment, those cancers that do get missed by all the hypothetical early detection schemes of the world, if we could identify exactly what’s wrong with a tumor using some of these profiling schemes which are so plentiful now, [one could develop] genetic or mutation-specific drugs. [This is] still horrifically expensive. If we can profile every tumor so that we can determine exactly which genetic or mutation-specific drugs are most effective for a particular tumor then we can in principle apply exactly the right drug to exactly the right patient. Of course, none of this works unless you have a health care system in which people have access to these materials without going completely under financially. So we as scientists, although we come from all over the political spectrum, have very strong feelings about the enormous importance of having these breakthroughs actually [be] available to people in a reasonable way.
The least expensive treatment might be prevention: can you talk briefly about that.
It’s been very interesting to understand at the molecular level what the nature of carcinogenesis is. For example in lung cancer, we now have a really clear understanding of why smoking causes lung cancer. We’ve known for decades that the way to reduce lung cancer incidents is for people not to smoke. If our high school teachers can do anything, anything in the discussion of cancer to keep teenage kids from getting hooked on cigarettes, it will have been the greatest possible thing and we’ll keep them alive to be cancer researchers. Many cancers though-breast, ovary, much of colon-develop despite their victims having done everything right. Breast cancer, for example, is very much the consequence of the normal life cycle of healthy modern women. Risk factors for breast cancer are related to hormones [in the body], they are also minimally related to the use of hormone replacement therapy. But most of the risk of breast cancer has to do simply with our being the most successful mammals there has ever been.
We are very good at reproducing at young ages and continuing to reproduce at what used to be old age, the forties. This means that the ductile cells of the breast, the cells that line the ducts that are responsible for lactation have the richest possible environment in which to live. They also have that environment if they’ve gone mutant. So breast cancer, in particular, is a consequence of a very healthy way of life. It’s not the desire of any of us who work on this disease to move our ways of life back to those of 17th-century women who had very little breast cancer. It is our desire to figure out what’s gone wrong and solve it at the level of the cell.
What would you say is where you see the biggest hope in this field?
To me, the very exciting future for cancer research will be our increasing capacity to couple what we learned at the level of genes, proteins, and pathways to schemes that will allow us to detect lesions before they are tumors that can be. And to develop drugs that are specific to particular types of genotypes of tumors. So the capacity to develop detection schemes and medicines that are very specific and therefore that work very well, work earlier, work better, and work in the case of treatments without the side effects that people now confront is where this field is headed.
It’s easily 20 years between a first, very relevant discovery and an end product that reaches a patient at a bedside. It’s remarkable to me and enormously heartening that this society understands that. That this society has been willing to put enormous amounts of resources into the work of a very large number of people with the expectation that collectively we will come up with answers. One consequence of that is that we’ve been able to recruit into cancer research–some of the brightest people of every generation over the last 30 years. That only works because of the quality of the teaching that these folks get when they are young. So thanks guys–keep it up.
If you look at scientific history in the past 200-300 years, would you say that the past 25 years have been remarkable and in what way?
We all hear that we are living through a revolution in science. But we really are living through a revolution in science! In the adult lives of those of us who are now working in the field, we’ve had a complete change in our understanding of what our basic makeup is all about. We now understand DNA. We understand what DNA does. We understand how it works. We understand how cells grow and divide. We understand what makes cells go wrong and consequently how cancer develops. In many ways it’s now putting the final, conceptual touches on that, allowing all of this information to now allow us to create complete catalogs, complete repertoires of what goes right during development and what can go wrong during development and to then build on that information to create compounds that allow us to detect what goes wrong and to correct it.
The idea of even saying this when I first started my career would have been laughable. But it’s true and people are doing it and within the professional lifetime of the students who we are now training I expect that we will have genotype-specific medications for many of the major cancers. That’s not very long.
What is your big dream for this work?
I would like for there to be no more mortality from breast cancer. I would like for us to be able to detect breast cancer at such an early stage that we can remove the offending cells with minimally invasive surgery at the level of what we would consider now as a needle biopsy. And a woman goes on about her life. That’s the scientific dream. My dream as a citizen is that we will have the economic common sense to make that kind of technology universally available when we’ve got it.
Do you have any parting thoughts for biology teachers?
We’re all teachers in this business. If you talk to anybody in the cancer field and ask them how did you get started doing what you’re doing, almost all of us will go back to a high school teacher. In my own case two high school teachers in math. It’s absolutely critical that youngsters feel empowered by their experiences as teenagers in math and science. I think teaching high school biology is the hardest job on the planet. The field moves incredibly fast. The amount of prep that a good high school biology teacher undertakes is just extraordinary. I don’t know how they do it. We very much appreciate and are in awe of what you all are doing at the high school level, thanks.